The collaboration of researchers from the Eötvös Loránd Research Network, the Eötvös Loránd University and the University of Pannonia has resulted a new publication in one of the world's leading multidisciplinary journals, PLOS ONE.
Attila Demény (member of the Hungarian Academy of Sciences, director of the Institute for Geological and Geochemical Research, ELKH Research Centre for Astronomy and Earth Sciences) and his research group discovered a peculiar phenomenon a few years ago concerning the stable isotope geochemical data of speleothems and the hydrogen and oxygen isotope ratios of their fluid inclusions. It was found that solutions enclosed in small cavities of speleothems do not retain the 18O/16O ratio characteristics of dripping water, but show significant 16O enrichment (Demény et al., 2016). The shift in the composition of the dripping water is caused by the precipitation of a previously unrecognized type of carbonate, amorphous calcium carbonate (“ACC”), which exchanges the 18O and 16O isotopes with H2O molecules in the entrapped solution during recrystallization to calcite (Demény et al., 2016). The next question was, what causes amorphous calcium carbonate precipitation and why it remains stable on the speleothem surface for weeks to months while amorphous carbonate precipitated under laboratory conditions transforms to calcite in minutes?
As the possibility arose that bacteria living on the surface of speleothems may be responsible for the precipitation of the amorphous material, researchers in earth science and biology started a collaboration. In the framework of the NKFI FK123871 project, Nóra Enyedi and Judit Makk, researchers at the Department of Microbiology of Eötvös Loránd University, and Péter Németh, researcher at the Institute of Materials and Environmental Chemistry of the Research Centre for Natural Sciences, examined in detail the mineralogical characteristics of bacterial carbonate. In addition to the morphological and structural analysis of carbonate, microbiological studies spectacularly show the stabilizing effect of the lipid-rich bacterial organic coating. The research team published the results in Scientific Reports of the Nature journal family.
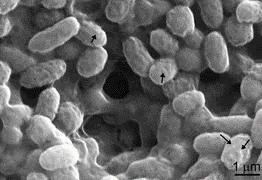
Carbonate precipitation on the surface of cultivated bacteria (black arrows)
Another aspect of the extensive research on cave carbonate formations is the geochemistry of “clumped isotopes,” that can be used to determine carbonate formation temperature. During carbonate precipitation from aqueous solution, the ionic species of dissolved carbon in solution (dissolved CO2, H2CO3, CO32–, HCO3–) are in dynamic equilibrium with each other. With the exchange of 18O and 16O isotopes, temperature-dependent thermodynamic equilibrium between the different species is expected. In contrast to thermodynamic equilibrium, however, the heavy isotopes of carbon and oxygen (13C and 18O) are preferentially clumped and the bonds are not broken due to the higher binding strength compared to light isotopes. The higher the formation temperature, the easier the break-up and the thermodynamic equilibrium to be reached. Researchers at the California Institute of Technology discovered the temperature dependence of heavy isotope clumping in 2006, establishing a new scientific field, “clumped isotope geochemistry”. The ELKH Institute for Nuclear Research in Debrecen (ATOMKI) has established a laboratory suitable for measuring clumped isotopes within the framework of a GINOP project (“IKER” project).

Thermo Scientific™ 253 Plus mass spectrometer and a Thermo Scientific™ Kiel IV automatic preparation unit at the Nuclear Physics Institute
The institutes began a collaboration to investigate Hungarian speleothems, and then, as a next step, to analyze the relationship between the bacterial carbonate precipitation described above and the clumped isotope compositions. The results obtained on speleothems differed significantly from the expected compositions for the given cave temperatures, suggesting that bacterial carbonate precipitation may have influenced the degree of heavy isotope clumping. Researchers in earth sciences and microbiology used the Baradla Cave as a natural laboratory, and with the permission and assistance of the Aggtelek National Park sampled and analyzed the carbonate precipitated on site. The essential condition of cave research is the participation of licensed cave research professionals, in this case Szabolcs Leél-Őssy, a lecturer at Eötvös Loránd University.

Germicide lamp and carbonate sampler in the Baradla Cave (Photo: Ágnes Berentés)
To demonstrate the effect of bacteria on carbonate formation, the sampling surface was illuminated with a germicide (cell-killing) lamp at a sampling point and a control point was kept untreated during the sampling period. The micromorphological characteristics of the collected carbonate samples differed drastically. In contrast to the irregular appearance of biogenic carbonate coated with biofilm, well expressed calcite crystals precipitated from the dripping water on the UV-treated surface. Clumped isotope compositions, in contrast, did not show a systematic relationship with UV treatment. The special composition of the studied speleothems is thus a consequence of the isotope fractionation processes taking place in the dripping water migration pathway, which provides essential information on the paleoclimatological applicability of the speleothems (see the research group's publication in PLOS ONE).
Research is continuing: genetic analyzes are being conducted to study the effects of cave bacteria, sampling is beginning in other regions of the world, and new scientific collaborations are already starting. The “NANOMIN” project of the Excellence Cooperation Program (KEP-1/2020), launched by the Hungarian Academy of Sciences and financed by the Eötvös Loránd Research Network in 2020, has resulted in a breakthrough in interdisciplinary research in addition to connecting research institutes and universities.

Carbonate with irregular morphology and covered by biofilm (left, control site) and abiogenic calcite crystals (right, UV-treated site).
Publications
Demény, A., Czuppon, Gy., Kern, Z., Leél-Őssy, Sz., Németh, A., Szabó, M., Tóth, M., Wu, Ch-Ch., Shen,Ch.-Ch., Molnár, M., Németh, T., Németh, P., Óvári, M. (2016a): Recrystallization-induced oxygen isotope changes in inclusion-hosted water of speleothems – Paleoclimatological implications. Quaternary International, 415, 25-32.
Demény, A., Németh, P., Czuppon, Gy., Leél-Őssy, Sz., Szabó, M., Judik, K., Németh, T., Stieber, J. (2016b) Formation of amorphous calcium carbonate in caves and its implications for speleothem research. Scientific Reports, 6:39602, DOI: 10.1038/srep39602
Demény, A., Rinyu, L., Németh, A., Czuppon, Gy., Enyedi, N., Makk, J., Leél-Őssy, Sz., Kesjár, D., Kovács, I. (2021) Bacterial and abiogenic carbonates formed in caves – no vital effect on clumped isotope compositions. PloS ONE 16(1): e0245621.
Enyedi, N.T., Makk, J., Kótai, L., Berényi, B., Klébert, S., Sebestyén, Z., Molnár, Z., Borsodi, A.K., Leél-Őssy, S., Demény, A., Németh, P. (2020) Cave bacteria-induced amorphous calcium carbonate formation. Scientific Reports 10, 8696.