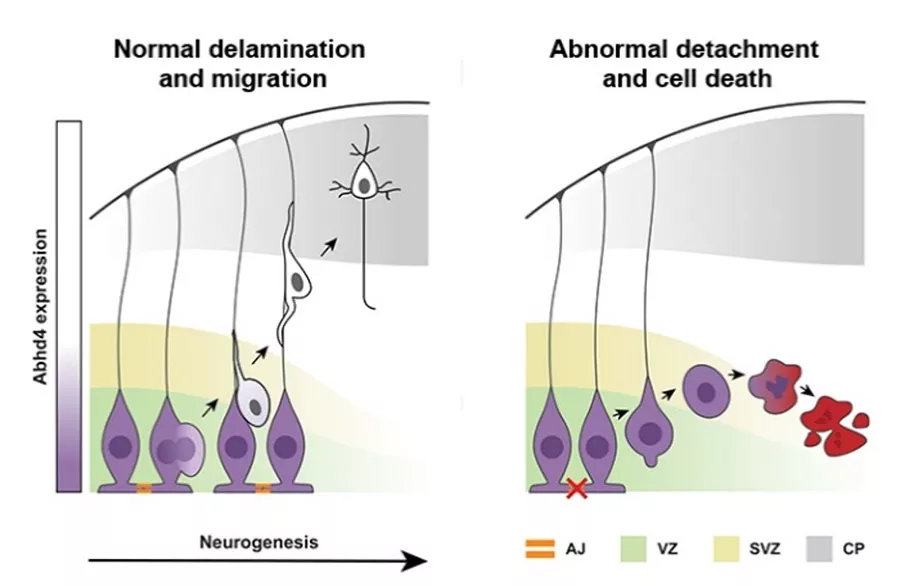
Epithelial-mesenchymal transition (EMT), a process initiated by the loss of cell to cell, and cell to extracellular matrix connections, is extremely important in both normal development and various diseases (cancer metastasis, fibrosis). In order to protect the organism from the dire consequences of inappropriate EMT, a specific form of apoptotic process named anoikis exists which is initiated by pathophysiological loss of the epithelial cell contacts to its surroundings. During mammalian cortex development, a process very similar to EMT happens during delamination of the fate-comitted daughter cells after asymmetric cell division. It has been so far unknown however, whether an anoikis-like protective mechanism exist to prevent the survival of abnormally delaminated and non-comitted proliferating progenitor cells: Equally importantly, it remained also unknown of how the normally delaminating daughter cells such as postmitotic neurons escape cell death.
The latest results of the Momentum Laboratory of Molecular Neurobiology offers a possible solution to both questions. We used in utero electroporation of a dominant-negative N-cadherin construct to force untimely delamination of the ventricular zone proliferating progenitors called radial glia cells. We demonstrated that disrupting the adherens junctions that connects these cells,we could indeed initiate a protective apoptotic process that we termed developmental anoikis. In addition, we showed that both acute and subchronic (3-day regime) maternal ethanol exposure, representing heavy and medium intoxication respectively, also elicited a similar protective apoptotic process in the proliferating zones of the embryonic cortex. Finally, we identified ABHD4, a serine hydrolase with an unknown in vivo function as a sufficient and necessary mediator of developmental anoikis.
See for details:
*László Z, *Lele Z, Zöldi M, Miczán V, Mógor F, Simon GM, Mackie K, Kacskovics I, Cravatt BF and Katona I (2020) ABHD4-mediated developmental anoikis safeguards the embryonic brain. Nature Communications, DOI: 10.1038/s41467-020-18175-4.
Képek
Az egészséges idegsejtek osztódás után felvándorolnak az agykéreg felső rétegeibe, miközben az ABHD4 enzim szintje lecsökken. A hibás sejtekben azonban az ABHD4 segítségével beindul az anoikis típusú sejthalál jelensége.
A kép készítője Balogh Kata
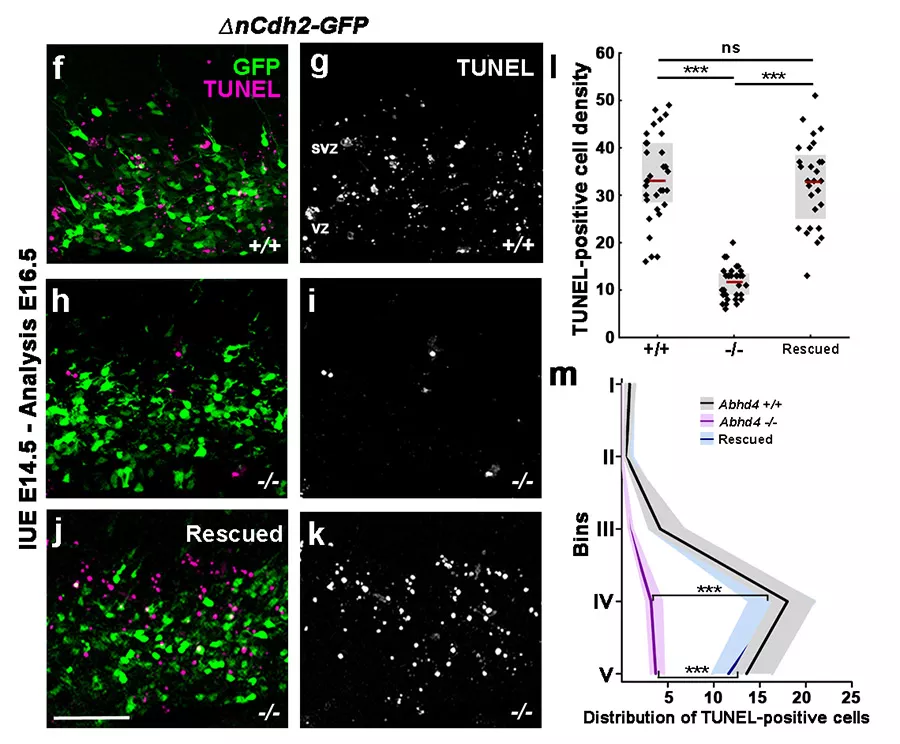
A domináns N-cadherin elektroporálása megnövekedett sejthalált okoz (f és g panel), ami az ABHD4 génkiütött állatokban nem jelentkezik (h és i panel). Az ABHD4 visszajuttatása a génkiütött állatokba helyreállítja a megnövekedett sejthalált.
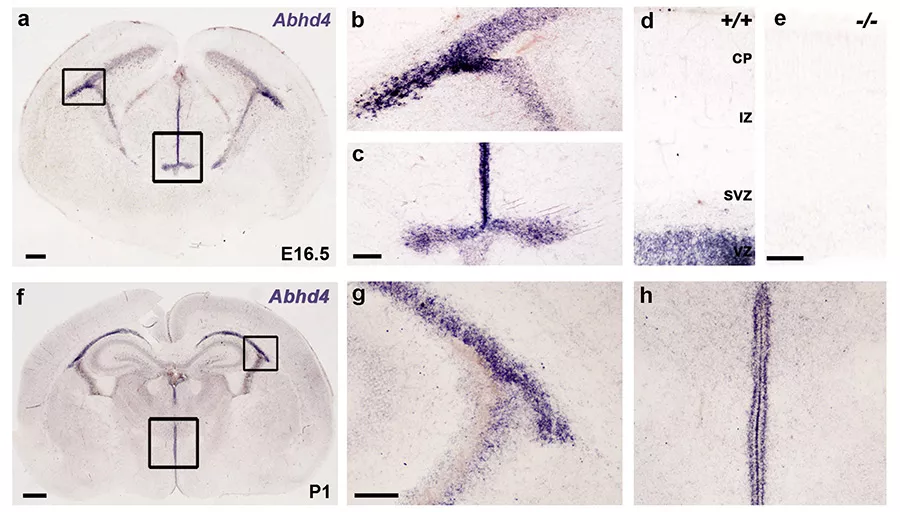
Az ABHD4 expressziója (lila színnel jelölve) az fejlődő agykéreg osztódó zónáiban. Az e. panelen, ami egy ABHD4 génkiütött állatból készült, látszik a jelölés specifikussága
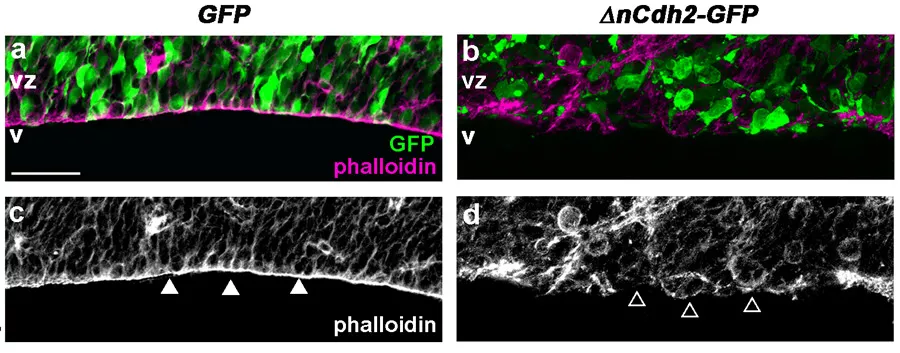
Az adherens kapcsolatok (lilával jelölve) lebomlása a domináns negatív N-cadherin (zöld GFP markerrel jelölve) elektroporálása után (b és d panel) a csak GFP elektroporálásához képest (a és c panel).
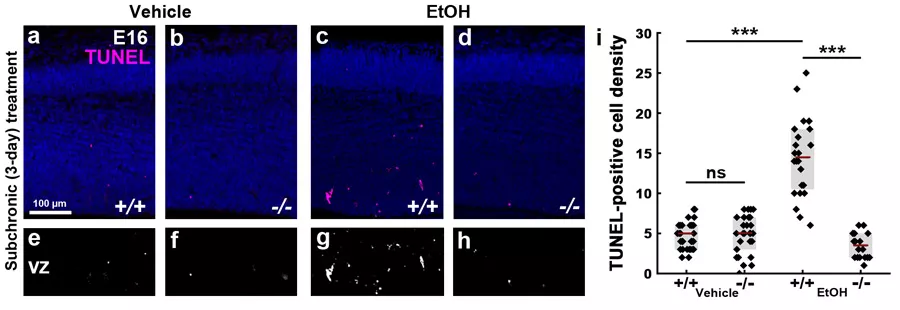
Az anyai alkoholfogyasztás megnövekedett sejthalált okoz a vad típusú embriók agyában (a és c panel). Ezzel szemben az ABHD4 génkiütött állatokban (d panel) ez a növedés nem látható), ami bizonyítja, hogy az ABHD4 jelenléte szükséges a sejthalálszint emelkedéséhez.
A teljes tanulmány angol nyelven a nature.com-on érhető el.