Three research teams from the Institute of Biochemistry at the Biological Research Centre in Szeged (BRC), led by Balázs Papp, Csaba Pál and Péter Horváth, have been investigating how new traits are created during the evolution of organisms. Through their experiments with yeast, they have shown that ecologically and clinically important new traits can emerge as a by-product of a deleterious mutation and secondary mutations that mitigate its deleterious effects. In doing so, the researchers at the BRC have uncovered a radically new evolutionary process in which deleterious mutations play a constructive role. Their results are reported in a paper published in the journal Nature Ecology & Evolution. The practical biotechnological significance of the discovery is that it opens up a new strategy for the laboratory cultivation of industrially useful proteins.
How do the characteristics of organisms appear and change during evolution? The molecular ‘raw materials’ for observable changes in visible properties are mutations, random changes in the hereditary material of organisms. There are countless examples of a new trait evolving as a result of the spread of beneficial mutations in a particular environment, whether it is the ability of bacteria to resist antibiotics or the ability of humans to decompose lactose. However, most of the mutations that are constantly emerging do not confer an advantage, but are detrimental to survival or reproductive chances. Could these deleterious mutations also contribute to the diversity of organisms? At first glance, you might think they could not, because even if a deleterious mutation changes certain traits, it will not spread due to its deleterious nature. This is indeed the fate of most deleterious mutations, but we know of cases in nature where they do not disappear, abut persist through another mutation that counteracts the deleterious effect. Such restorative mutations are called compensatory mutations, and the process itself is called compensatory evolution. If this can result in new organisms with altered characteristics, it suggests that deleterious mutations can also contribute to the diversity of the living world. The BRC researchers were looking for an answer to the fundamental question of whether new properties are actually created in this way.
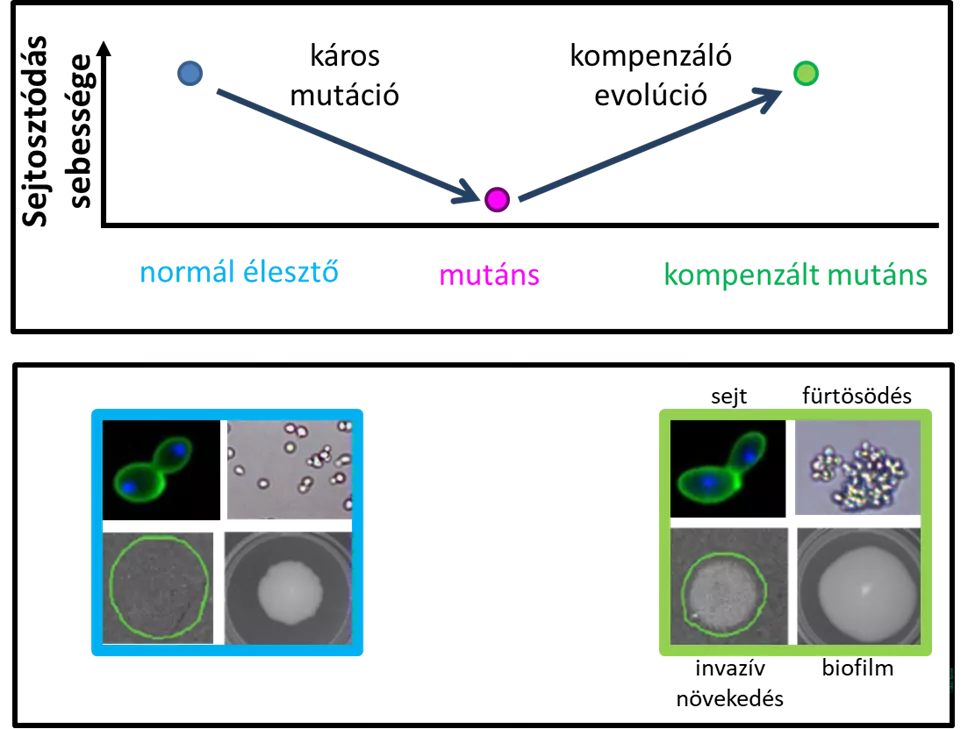
For their experiments, they used budding yeast, which is familiar from the kitchen. Single-cell budding yeast is often used in laboratory evolutionary experiments because it is easy to grow and capable of doubling itself by cell division in an hour and a half, thus creating a new generation. In a previous study, the researchers at the BRC have already found that compensatory evolution can easily take place in budding yeast. As a starting point, the deleterious mutations were artificially and individually engineered by the complete deletion of a single gene, resulting in a collection of hundreds of mutants in which the yeast division rate was significantly decelerated. The mutant yeast cells were then grown in the lab for three months, waiting for compensatory mutations to emerge, which they did, and as a result, growth rates were restored in the majority of the mutants.
In their latest experiment, the researchers wanted to find out how often the resulting compensated mutants differ in some way from the so-called wild yeast that did not carry the mutations. Several properties were investigated, and as laboratory budding yeast is typically unicellular, changes in cell shape were first observed using automated microscopic image processing. In the study, it was found that the compensated mutants often have cell shapes different from wild-type yeast. In fact, the types of changes observed and their magnitude were very similar to those observed in naturally occurring budding yeast variants.
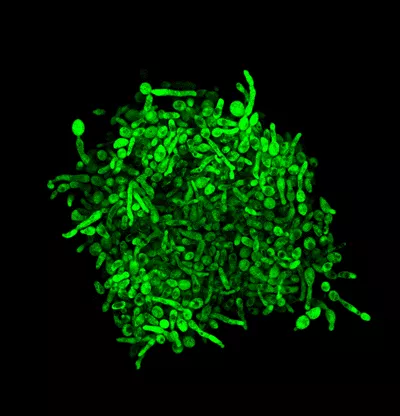
Therefore, the cell shape often changed, but the researchers also wanted to see if the yeast cultured in the experiment could also form multicellular shapes. Although multicellular forms are common in nature, laboratory budding yeast has not yet been shown to be able to form this shape. This issue is of particular importance because, in pathogenic yeast species, multicellular forms facilitate host attack.
To answer this question, the researchers have now investigated the emergence of three different forms of multicellularization – clustering, biofilm formation and invasive growth. During clustering, the imperfect division of yeast cells means that the cells growing in the liquid do not separate. In biofilm formation, cells can adhere to each other and to solid surfaces such as medical equipment, which can cause serious health issues. During invasive growth, cells can penetrate solid surfaces such as animal and plant tissues using thread-like formations. Surprisingly, several of the compensated mutants developed biofilms larger than those in the wild yeast, while in others clustering or invasive growth emerged as a completely new trait. This is remarkable because these traits have not been selected for in laboratory evolution, so they are merely by-products of compensation for deleterious mutations.
BRC researchers were the first to show that the combination of deleterious mutations and compensatory evolution can lead to the emergence of ecologically and clinically important traits. This discovery also suggests a new evolutionary picture, showing that evolution is not just a straight-line ‘triumphal process’, as deleterious mutations are an important initial step in evolutionary adaptation. Further research may answer the question of how often this process occurs in nature. However, the recent discovery also has practical significance as an important branch of biotechnology uses the principles of natural selection to cultivate proteins with new properties in the laboratory. Mutations that are detrimental to the function of proteins, in combination with other beneficial mutations, can promote the formation of qualitatively new enzymes.